How Incandescent Lights Work
- Heat a filament by running electrons through it. Typical
temperatures are around 5000K. So the radiation peaks at:
Wavelengthmax = 0.29/5000 = 0.000058 cm = 5800 Å
- What kind of spectrum do you expect?
A Planck curve.
How Fluorescent Lights Work

- Turn on the power, start boiling electrons off the filaments and send them
flying back and forth through the tube at 60Hz.
- The electrons crash into atoms, collisionally excite the electrons
in the atoms to higher levels. When the electrons in the atoms drop back down
to lower excitation states, photons are emitted - for most tubes, these are
primarily UV photons.
- With no phosphor coating, you get a "black light".
- With a phosphor coating, the UV photons are absorbed by the atoms in the
phosphor and excite them to high levels. The de-excitation occurs through a
cascade of photons where there area number of visible light photons for each
UV photon absorbed.
Fluorescence and fluorescent lights
- Don't emit in the IR ("cool" and energy efficient).
- Do emit in the UV (this is why the shoulders in your clothes fade).
- Are better for plants as they mimic the solar spectral coverage better.
- "Whiter-than-white" detergents leave a residual of phosphor-like
material on your clothes that absorb UV from the Sun and re-emit blue photons
to make the balance of light reflected seem whiter
- The "decay time" for some of the downward atomic transistions can be
quite long. That's a good thing, or glow-in-the-dark toothbrushes would not be possible.
- The are many examples of fluorescence in astronomy.
The picture below is of the expanding shell of gas that was expelled
in a supernova explosion (much more about these later. A spectrum of
this object would show emission lines from oxygen, nitrogen and
sulphur.

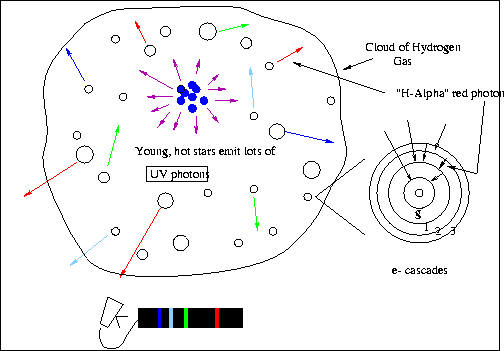
- The ``HII region" illustrated below this is a ``radiationally excited''
example of fluorescence in astronomy. Young, hot, massive stars produce
lots of UV photons that ionize the cloud of gas that the stars formed
from. When electrons in the plasma recombine, the electrons cascade
down through the energy levels giving off visible-light photons.
One of the most likely downward transitions is when and electron drops
from the second to the first excited state. This produces a photon
at 6563 angstroms -- we see this as red light.
- Here is a real version of an HII region -- the ``Rosette Nebula''.

- As we will see later, at the end of the life of a star like the Sun, the outer
layers of gas will expand into space. This gas will be bombarded by UV photons
from the hot stellar core that is left behind and through the process of fluorescence
the nebula will emit the red ``H-alpha'' line along with an emission line
in the green part of the spectrum that comes from oxygen atoms. The Catseye Nebula
is an example.

- The Aurora of the Earth is an example of a "collisionally" excited gas
that radiates. Here, charged particles streaming from the Sun
spiral along the Earth's magnetic field lines to the poles and
crash into atoms in the upper atmosphere. The atoms are knocked into excited
states and emit photons as the electrons in the atoms drop back to
lower energy levels.







