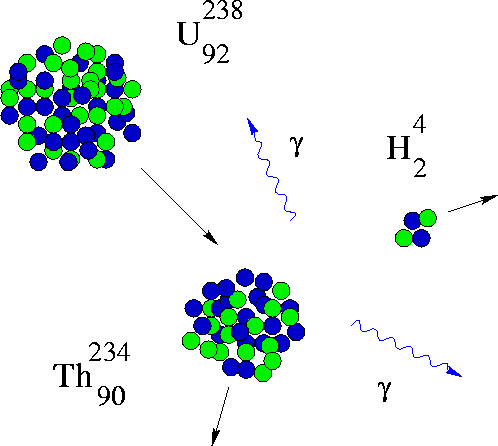
For the "heavy" elements in the Periodic Table, there are some that have nuclei that spontaneously fall apart - these are the radioactive elements. The wild thing about this is that if you weigh the initial nucleus and weigh the bits after the "fission", they don't add to the same total.
This equation says that Mass and Energy are interchangable.
Take the mass difference between the Uranium nucleus and the sum of the Thorium and Helium nuclei, multiply by the speed of light squared and you have the amount of energy that was converted from mass in the reaction (released in the form of gamma rays).
 mc2
mc2
where m is the mass difference between the Uranium nucleus and the fission product nuclei.

 2H1 + e+ +
o
2H1 + e+ +
o

 as a nuclear-fusion powered object?
as a nuclear-fusion powered object?